Loss of microstructural integrity and connectivity in the white matter, appearance of white matter hyperintensities (WMHs), morphological changes, and functional brain activity disturbances have been described in the brain imaging of premutation carriers with FXTAS.
WMHs
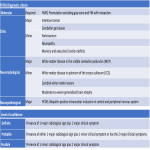
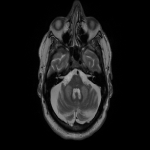
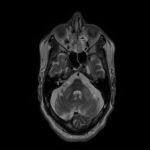
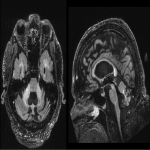
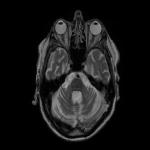
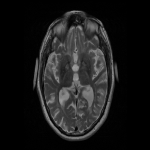
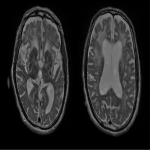
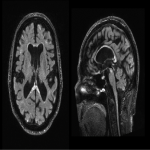
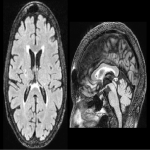
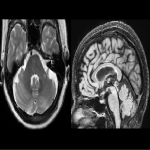
WMHs serve as the major radiological hallmarks of FXTAS: Bilateral hyperintensities of the middle cerebellar peduncles (MCP sign) on T2-weighted and FLAIR MRI are found in 52-83% of men and 0-13% of women with FXTAS. Hyperintensity in the splenium of the corpus callosum (CCS sign) has also been described and occurs similarly in males (65–72%) and females with FXTAS (50–60%) [4], [5], [6], [7]. (Figure 3-4-5-6-9-10-11-12) Interestingly, the presence of both the MCP and CCS signs is much more frequent in FXTAS (41%) than in other neurodegenerative diseases including multiple system atrophy-cerebellar type (0%), essential tremor (0%), Parkinson’s disease (0%), Alzheimer’s disease (0%), and stroke (3%) [6].
The MCP sign is associated with more severe cognitive deficits and a longer history of symptoms [8]. Even though the MCP sign is highly specific to FXTAS, it is not pathognomonic and can also be found in premutation carriers without clinical symptoms of FXTAS [9] and other neurodegenerative disorders [4].
WMHs on T2-weighted and FLAIR MRI are commonly observed in FXTAS in both cerebral and cerebellar white matter, showing significantly higher volume in FXTAS compared with age-related controls and carriers without FXTAS [5].
Hyperintensities are also commonly observed in pons, insula and in the bilateral pallidal region in FXTAS [10], [11], [12]. (Figure 3-4-7-8)
Structural changes
Generalized brain volume loss has been described in patients with advanced FXTAS; in the entire cerebrum and cerebellum, brainstem and cortical and subcortical regions. Ventricular enlargement is also present in FXTAS as well as thinning of the corpus callosum [11]. However, longitudinal studies have shown different patterns of atrophy progression in each of these regions.
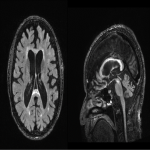
There is a progressive ventricular enlargement within the 4 ventricles as FXTAS develops and a deleterious cycle between ventricular enlargement and brain deformation that may commonly occur during aging and FXTAS progression but becomes accelerated in FXTAS [13]. Large cavum septum pellucidum and cavum vergae (spanning ≥ 6 mm in the coronal direction) have been reported in about 13.9% premutation carriers [14]. (Figure 10)
Atrophy and diffusion-weighted imaging signal loss in the subcortical structures : thalamus, caudate nucleus, putamen, and globus pallidus have been described in carriers with FXTAS compared to controls, and importantly, many of the measurements exhibited robust correlations with symptom severity [15].
The cerebellum and brainstem are likely affected during both neurodevelopment and FXTAS-associated neurodegeneration. FXTAS patients exhibit cerebellar atrophy and accelerated brainstem atrophy compared with controls and the premutation carriers without FXTAS [5] [16] [17] [14]. (Figure 3-4-5-6-7-9-10 ) MCP width was reduced in carriers who developed FXTAS in subsequent visits as well as cross-sectional area of the midbrain and pons that were reduced in non-FXTAS premutation carriers compared with controls. Reduced MCP width and reduced brainstem cross-sectional areas have been proposed as potential risk markers for developing FXTAS [18]. (Figure 5-6)
Increasing CGG repeat numbers correlate with a greater degree of cerebral and cerebellar atrophy in male carriers, ventricular enlargement, and whole-brain white matter hyperintensity volume. In females with FXTAS, the magnitude of cerebellar volume loss and the severity of white matter disease is generally less than that in males [11] [5].
Structural connectivity:
Diffusion tensor imaging (DTI) has shown abnormalities in connectivity in patients with FXTAS. Premutation carriers with FXTAS showed reductions in structural connectivity in motor, limbic, association and callosal fiber tract categories. Additionally, superior cerebellar peduncle tract volume correlated negatively with CGG repeat length in premutation carriers both with and without FXTAS. Patients with FXTAS showed significant reductions in fractional anisotropy in numerous white matter tracts, especially the MCPs, the superior cerebellar peduncle, the cerebral peduncle, the fornix and the stria terminalis [17]. Reduced structural connectivity showed significant associations with behavioural and attentional self-regulation and dexterity [19].
Functional Imaging:
Progress has been made in understanding neuronal substrates of motor control and emotional processing using functional MRI in premutation carriers.
Cerebellar pathology and visual-spatial processing pathology have been described in FXTAS and pre-symptomatic premutation carriers; functional connectivity alterations of cerebellar-cortical networks during sensorimotor behavior have been proposed to represent a "prodromal" feature associated with FXTAS degeneration [20].